FERTILITY ROAD ARTICLE
View From the Embryology Laboratory
An embryo’s journey in the laboratory by Anya Hein
Anya Hein, one of seven embryologists in the ten embryology team at Oregon
Reproductive Medicine shares some insights on the critical time that patients’ embryos spend growing and developing in the laboratory.
 When choosing a fertility provider, picking one that you trust and feel comfortable with is an essential part of the process. Patients typically focus on their individual doctor, but they do not always get to know more about the embryology laboratory and the embryology team that will be working on their behalf when selecting a fertility provider. When patients learn more about the IVF process, they realize the extremely crucial role that the embryology lab and team will play in their journey to parenthood. Every laboratory and embryology team has its unique attributes which can contribute to success, so it is helpful for patients to get to know more about this part of their IVF team and have trust in them as well.
When choosing a fertility provider, picking one that you trust and feel comfortable with is an essential part of the process. Patients typically focus on their individual doctor, but they do not always get to know more about the embryology laboratory and the embryology team that will be working on their behalf when selecting a fertility provider. When patients learn more about the IVF process, they realize the extremely crucial role that the embryology lab and team will play in their journey to parenthood. Every laboratory and embryology team has its unique attributes which can contribute to success, so it is helpful for patients to get to know more about this part of their IVF team and have trust in them as well.
As an embryologist, “Guardian of Embryos”, I know that it can seem as though we want to keep all the magic that happens in our little laboratory to ourselves. There tends to be a veil of mystery over what is going on in the IVF laboratory. As a patient you probably won’t meet one of us as you begin your cycle and you may not even meet us on the day of your egg retrieval. A patient might wonder; are there even any embryologists in there? What are they doing with my gametes? It is a significant leap of faith that we ask our patients to have immeasurable trust in us as we care for their hopes of becoming parents.
The IVF process demands a level of emotional stress and immense patience most of us have yet to experience. With every stage, we ask the patients to wait, wait again, and then wait again. Perhaps a little insight into the process could make the heart feel just a little lighter and ease the burden of all that waiting. It is important to remember that every clinic is different and their success rates may be influenced by many factors, not just the quality of their embryology laboratory and team. However, the general techniques and flow of the embryos within every laboratory will be very similar.
Preparation for Day 0
A patient, either a woman looking to conceive with her own eggs or an egg donor, will for about 10-12 days be on specific medications that are designed to stimulate her ovaries to produce as many eggs as possible. Every woman has a cohort of available “pre-eggs” each month, known as follicles. While naturally only one or two of these follicles will grow into a mature egg and ovulate, the stimulation will allow most of these available follicles to grow, mature, and be available for insemination in the laboratory. Once the follicles have grown to a desirable size, the patient will be “triggered” for egg retrieval, which is known as Day 0. The gametes (sperm and eggs) will be in the embryology laboratory from Day 0 through Day 6 (blastocyst stage).
Day 0
On the day of egg collection, the eggs and the sperm will come together. This can happen in two different ways, intracytoplasmic sperm injection (ICSI) or standard insemination.
ICSI involves the removal of the pillow-like cumulus cells using an enzyme called Hyaluranidase in order to visualize the maturity of each egg prior to injecting the sperm. At our clinic, approximately 90% of the cases will utilize ICSI to maximize the number of fertilised eggs and help combat low sperm count and/or motility.
The levels of maturity of the egg can be seen down the microscope once the cumulus cells have been removed. The three degrees of maturity are defined as Germinal Vesicle (GV), which is the most immature, Metaphase I (MI or GVBD), and Metaphase II (MII or PB).

Only when an egg appears to have a Polar Body (MII stage) present can they be fertilised. It is very helpful that eggs show us when they are mature so we know which ones are ready to accept a sperm and possibly fertilise. On average, 75% of the collected eggs will be mature.
It is important to realise that everything that happens in the lab and in nature is a process, not a moment. When we perform ICSI, it is not instantly known whether those eggs will fertilise. We begin ICSI and inseminations after lunchtime and it is one of the last things we do in the lab for the day. We inject the eggs, whisper our good intentions and tuck them away in their warm incubators for the night. Applying accurate timing and vigilant patience, we have faith that the process will continue and the magic will happen between those eggs and that sperm.
Standard Insemination involves the eggs retrieved being divided among different drops of culture media and then inoculated with clean, motile sperm. These eggs will not be stripped of their cumulus cells and therefore the maturity of them will not be known until the following day, at fertilisation check.
Days 1-3
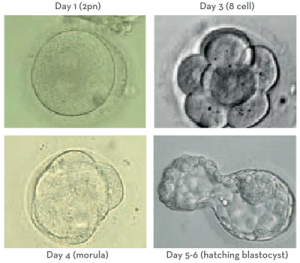 On Day 1, approximately 16-18 hours after we introduced sperm to the eggs, we check to see how many have fertilised, separating the fertilised eggs from the unfertilised and the abnormally fertilised.
On Day 1, approximately 16-18 hours after we introduced sperm to the eggs, we check to see how many have fertilised, separating the fertilised eggs from the unfertilised and the abnormally fertilised.
Fertilisation is only visible to our eyes for a few hours before the embryo will begin to divide. For those eggs that were inseminated with standard insemination we must first remove the cumulus cells before we can see fertilisation. Hyaluranidase is a naturally occurring enzyme in sperm, so these eggs are now easily separated from their cumulus after an evening with a lot of sperm.
When we look down the microscope we can see two small spheres in the eggs; which designate the normally fertilised ones from the rest. These small spheres are called pronuclei; we look for two of them (2pm). When we see two we know that there is both paternal DNA (sperm) and maternal DNA (egg) present.
We expect about 75-80% of the mature eggs to fertilise normally. Here again, we see a reduction of eggs from the number we started with; generally fewer eggs than follicles, fewer mature eggs than total eggs and now fewer fertilised eggs (rygotes) than inseminated eggs. There are many reasons eggs do not fertilise. There can be an egg factor due to age or quality, or sperm factors that prevent the eggs from fertilising, or potentially a combination of both egg and sperm.
After checking for fetilisation we want to put them back in their incubator as quickly as possible. IN there they thrive and grow. We leave them to themselves until Day 3.
Day 3-4
On Day 3, we assess the embryos’ growth to this point. If Comprehensive Chromosome Screening (CCS) is planned then we will also hatch a small hole in zona pellucida (shell) of each embryo. Embryos will naturally hatch out of their zonas prior to implanting in the uterus, so it is a common part of their progress; we just give the embryos to a little early nudge.
This assisted hatching will help to make the biopay process easier on Day 5/6. About 70-80% of the cases at our clinic utilize CCS. When we take the embryos out and move them into the appropriate media for this stage of growth we grade them based on the number of cells, percent fragmentation, and symmetry.
We want the embryos to be around 6-8 cells by this day. The embryos are beginning to distinguish themselves from each other at this stage. About 95% of the initial embryos will continue to grow to Day 3.
Days 5-6
 The next time we look at the embryos is on Day 5. Without disruption, between Day 3 and Day 5 the embryos make the very difficult transition from a multicellular cleavage stage embryo
The next time we look at the embryos is on Day 5. Without disruption, between Day 3 and Day 5 the embryos make the very difficult transition from a multicellular cleavage stage embryo
to a morula. Then by Day 5/6, they make the transition from a morula to a blastocyst.
The blastocyst is the stage at which differentiation begins. At the morula stage, the embryo’s cells compact together and decide which cells amongst them will continue to divide and become the Inner Cell Mass (ICM) and which will become the trophectoderm. These are the crucial parts we look for in the embryo at the blastocyst stage. The ICM is the small grouping of cells within the embryo that become the baby and the trophectoderm is the honeycomb-like cells surrounding the ICM that will become the placenta.
Once they have reached this stage, the laboratory environment becomes unsuitable for further growth. Thus Day 5/6 are the days at which these embryos are either transferred back into a uterus, vitrified (a method of rapid cooling of the embryo, necessary for prolonged suspended storage), or biopsied for CCS testing and then vitrified. As with all things in nature, not all embryos grow at the same rate. There is a range of growth that is acceptable; therefore, we look for the same signs of viability and growth on Day 6 as we do on Day 5.
Due to the high level of energy and “perfection” required to grow from a cleavage stage embryo to a blastocyst, on average, we observe only about half of the viable Day 3 embryos develop to Day 5/6. Some embryos may grow to the blastocyst stage but are not of strong enough quality to be transferred or biopsied.
There can be many reasons why the embryos do not continue to grow to Day 5/6; sperm factor, egg factor, lack of sufficient mitochondrial energy, or chromosomal abnormalities, the list really goes on and on. Whatever the factor may be, when we see eggs drop-out and then embryos drop-out there is an abnormality within the egg or embryo preventing it from continuing in the process. Even with the perfect conditions, they may lack the internal mechanisms to continue to thrive. And it is very important to remember these embryos would not make a baby.
Throughout the IVF process, one must remember that follicles do not equal eggs, eggs do not equal zygotes, zygotes do not equal blastocysts, and blastocysts do not equal pregnancies. Every cycle will be different and it can be extremely frustrating for patients to have little control. It’s helpful to enter into treatment with reasonable expectations, a basic understanding of the embryology process, and a view of the bigger picture. The end goal being a healthy baby, and each healthy baby requires only one normal embryo that has had a caring start.
Rest assured that no matter how long or winding your journey, your embryology team will be there ready to steady you through any moments of extreme emotion, anxiety, and potential heartbreak on your road to parenthood, while caring for your embryos with a skilled hand and tender heart.
More information can be found on the Oregon Reproductive Medicine website oregonreproductivemedicine.com.



